Our Ambition Is Quite Simple: To Be the Best
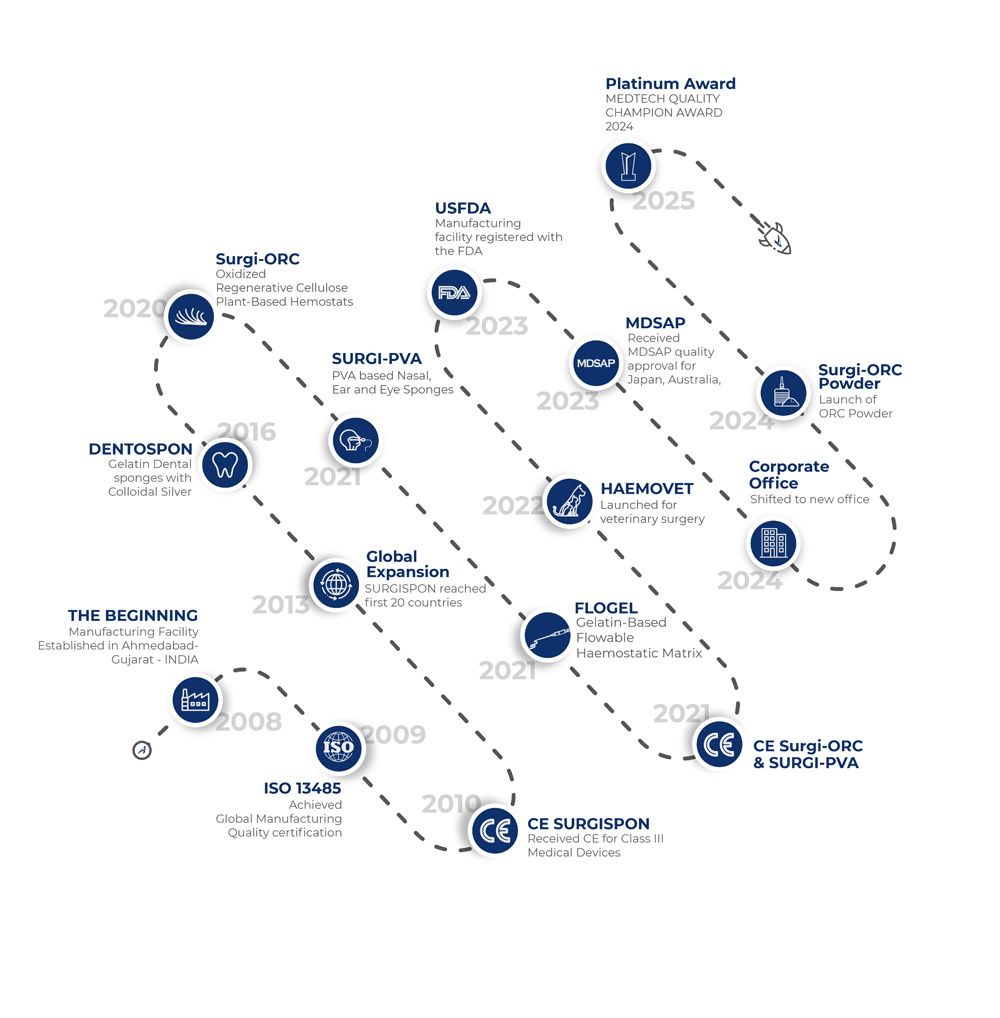
Aegis Lifesciences, established in the year 2008 is world’s leading manufacturer& exporter of all major types of Haemostats – made of Absorbable Gelatin Sponges and Haemostatic powder, Oxidized Regenerated Cellulose (ORC), medical grade PVA sponges and Flowable Haemostatic Matrix.
It leads the industry with its own brand names.
- SURGISPON® for all Gelatin Sponges, covering all types of Surgeries like General Surgery (Sponges), Cardiac (Patches), ENT, Dental, Proctology (Anal Tampons) etc.
- Surgi-ORC® for Oxidized Regenerated Cellulose, covering all variants like Original, Knit, Fibril and Non-Woven
- SURGI-PVATM for medical grade PVA dressings/sponges covering Nasal & Ear packs, Eye Spears & Drains
- DENTOSPON® for Dental absorbable sponge with colloidal silver
- FLOGEL® Flowable Gelatin Haemostatic Matrix Kit for tight and irregular spaces especially at a difficult to reach site
- HAEMOVET® Absorbable Gelatin sponges for all types of Veterinary surgeries
Aegis offers products in more than 80 countries, including the most quality-conscious markets like Europe, USA, UK (NHS), Latin & Central America, CIS including Russia, Middle East, Africa, and Southeast Asia regions.
The most promising fact about the Company is its ability to reach the zenith of success in few years. Not to mention, the base of its operations relies on international quality standards along with the state-of-the-art in-house manufacturing facilities. Aegis facility earned top certifications in the area such as ISO:13485, MDSAP, US FDA and GMP standards of many countries.
Aegis Lifesciences has gained international certificates of CE (Class III medical devices) & ISO: 13485 Quality Assurance System from DNV GL Nemko Presafe AS, Norway.
Also, being truly customer-centric in thought and action, Aegis believes that customization and quality is the key for creating and retaining a sturdy customer base. The USP of AEGIS has always been it offers BEST quality products at the BEST prices.


Why us?
Our history