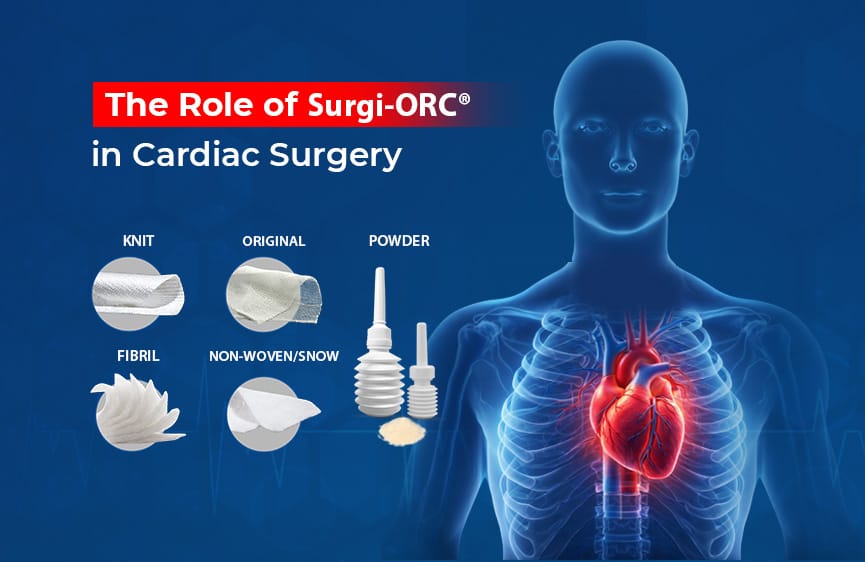
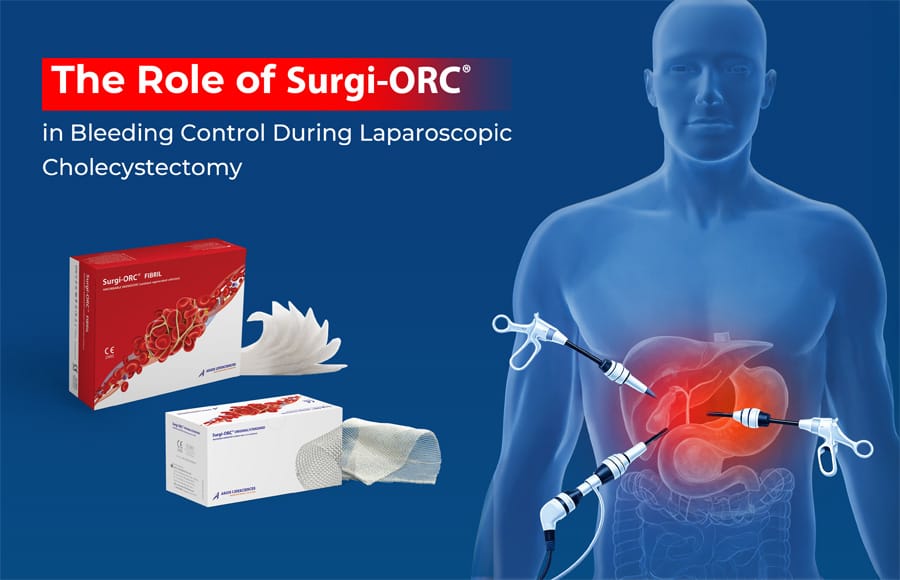
Fibrillar Absorbable Hemostat: A Promising Aid for Bleeding Control in Colorectal Tumors

Colorectal cancer, one of the most prevalent cancers worldwide, often requires sophisticated treatment modalities to achieve optimal outcomes. Among these, endoscopic submucosal dissection (ESD) has emerged as a promising technique for the early-stage management of colorectal tumors. However, the procedure’s success can be tempered by postoperative complications such as delayed bleeding and perforation, as well as the challenge of managing large mucosal defects. Moreover, exposure to gastrointestinal flora increases the risk of bacteremia and endotoxemia. In the quest for safer and more effective solutions, researchers and clinicians are continually exploring alternative approaches. Fibrillar absorbable hemostat such as Surgicel® Fibrillar™ (Ethicon), however, emerges as a game-changer offers a multifaceted approach to post-ESD care, providing both hemostatic support and localized bactericidal effects.
A notable product in this realm is Surgi-ORC® Fibril from Aegis Lifesciences, featuring the following key characteristics:
Key Features of Surgi-ORC® Fibril:
- Lightweight, tufted, soft and layered structure
- Provides high flexibility and conformability to surgical sites
- Suitable for irregular shaped wounds and hard-to-reach areas
- Bactericidal properties
- Soft, layered material conforms and “melts in” to bleeding tissue
- Completely absorbable
- 7 layers fibrillose fabric

- Designed for hemostasis in

How does Surgi-ORC® Fibril Work?
Surgi-ORC® Fibrilis composed of oxidized regenerated cellulose, which, upon contact with blood, swells into a gelatinous mass, aiding in clot formation and preventing further bleeding. Moreover, its low pH of 3.4 exhibits potent bactericidal properties, reducing the risk of infection significantly.
Clinical Study Results:
A clinical study was conducted at the University of Soonchunhyang Hospital in Bucheon, Korea, sheds light on the effectiveness of Surgicel® Fibrillar™ in colorectal ESD. In a cohort of 49 patients, Surgicel® Fibrillar™ was successfully applied to cover the entire mucosal defect following ESD, without any Surgicel-related complications. Remarkably, none of the patients in the Surgicel group experienced delayed bleeding or perforation, marking a significant improvement over conventional ESD techniques. Moreover, Surgicel® Fibrillar™ was associated with shorter hospital stays and lower inflammatory markers, indicating quicker recovery and improved postoperative outcomes.

Fig.1: A) Lateral spreading tumor; B) Dissected colon LST by colon endoscopic submucosal dissection.; C) Surgicel was placed onto the mucosal defect to cover the entire mucosal defect; D) Surgicel swelled into a gelatinous mass [1].
Conclusion:
In the realm of colorectal cancer treatment, every advancement brings us closer to our ultimate goal: improving patient outcomes and enhancing quality of life. Fibrillar absorbable hemostat represents a pivotal milestone in this journey, offering a potent combination of hemostatic efficacy and antimicrobial protection. As we continue to push the boundaries of innovation, let us embrace the transformative potential of Fibrillar absorbable hemostat and embark on a path towards safer, more effective colorectal endoscopic resection.
References:
[1] Myung YS, Ko BM, Han JP, Hong SJ, Jeon SR, Kim JO, Moon JH, Lee MS. Effectiveness of Surgicel® (Fibrillar) in patients with colorectal endoscopic submucosal dissection. Surgical endoscopy. 2016 Apr;30:1534-41. DOI:10.1007/s00464-015-4369-5